Neditol® XL meets all the relevant regulatory requirements of a bioequivalent medicine (reference product Detrusitol® (tolterodine) XL 4mg prolonged release capsules).1 Three bioequivalence clinical trials were performed comparing Detrusitol® (tolterodine) XL (UpJohn UK Limited). Two single dose studies were performed under fed and fasted conditions and a steady state study was performed under fasted conditions.1 The graphs are shown below.1
Single dose Study Comparing Neditol XL 4mg and Detrusitol XL 4mg Under Fasting Conditions1
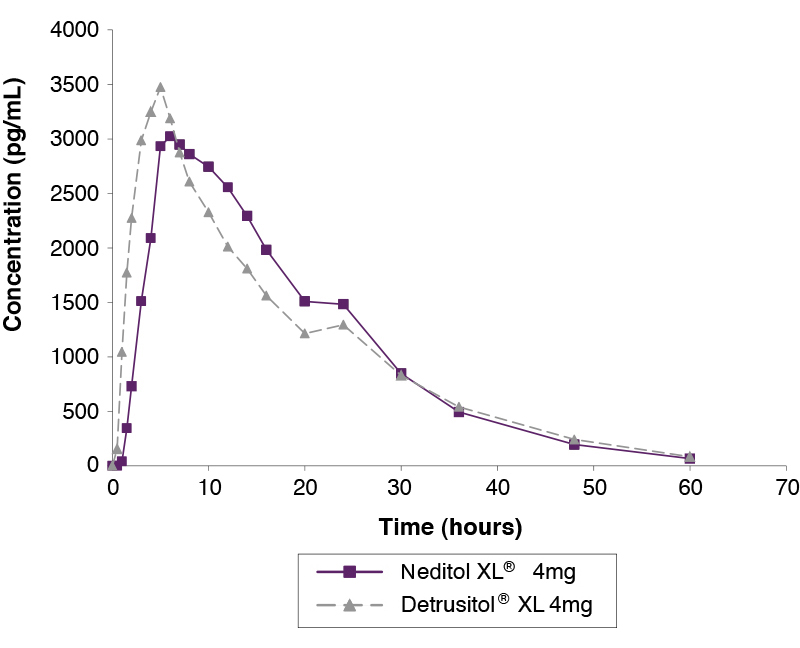
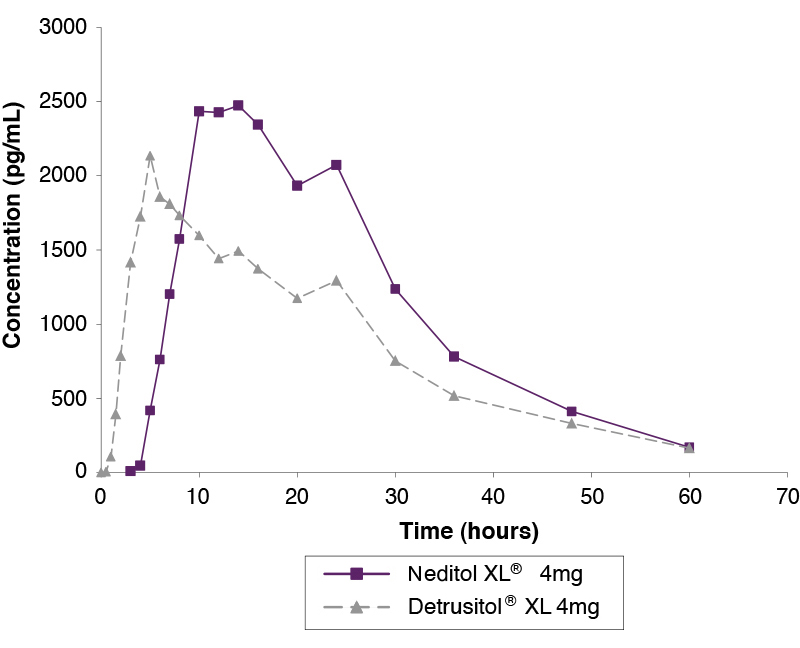
Single Dose Study Comparing Neditol XL 4mg and Detrusitol XL 4mg Under Fed Conditions1
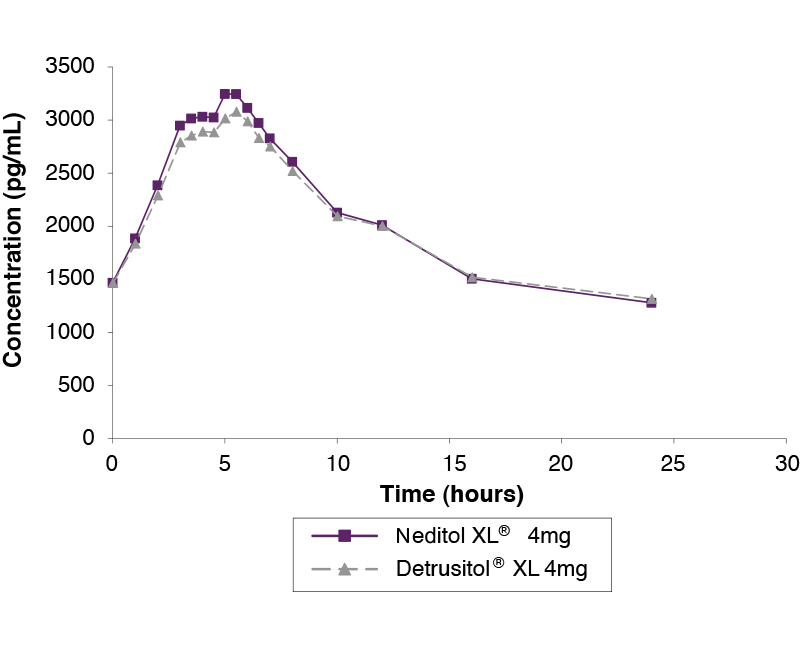
Steady state study comparing Neditol XL 4mg and Detrusitol XL 4mg under fasting conditions1
 Reference: 1) Data on file. 1010067149 v 6.0 September 2023
Reference: 1) Data on file. 1010067149 v 6.0 September 2023
Adverse events should be reported. Reporting forms and information can be found at www.mhra.gov.uk/yellowcard.
Adverse events should also be reported to Aspire Pharma Ltd on 01730 231148
For more information about Neditol XL, please see the abbreviated prescribing information.














